|
The ‘Cutadapt_mqc-generalstats-cutadapt-percent_trimmed’ is twice (around 1.4) when I provide the adaptor sequence as compared to when no adaptor sequence is specified (around 0.8 to 0.9 )as a parameter. I tried both methods (1) inserting adaptor sequence and (2) not providing any specific information for the adaptor, and got different results on the MultiQC of the trimmed reads. It seems totally normal that the adapter content would go away after this step. So I decided to run this trimgalore / cutadapt step. Using one of the supplied Fasta Files Illumina adapter and other technical sequences are copyrighted by Illumina,but we have been granted permission to. It seems that I have the 'Illumina Universal Adapter' in my reads and I need to trim the adapter sequence using trimmomatic. param-files “FASTQ/A file #2” : both _2 fastqsanger datasets (multiple datasets)The order is important here! In terms of adapter content- this is what started it all, I saw FastQC return Illumina Universal Adapter content at multiple positions in the original reads, increasing all the way up to the read end. I ran the fastqc quality check on the fastq sequencing file.param-files “FASTQ/A file #1” : both _1 fastqsanger datasets (multiple datasets).“Single-end or Paired-end reads?” : Paired-end.Ith the following parameters to trim low quality sequences: TUTORIAL 2 PARAMETER FOR CUTADAPT (EXTRACTED) param-text “Enter custom 3’ adapter sequence” : AGATCGGAAGAGCACACGTCTGAACTCCAGTCAC.param-text “Enter custom 3’ adapter name (Optional)” : Illumina. 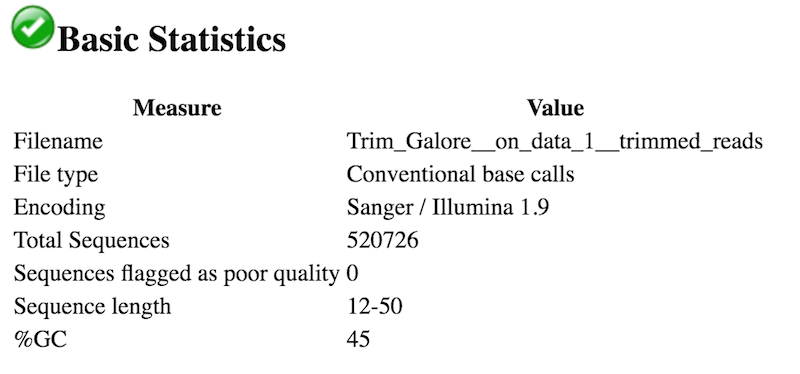
param-select “Source” : Enter custom sequence.TUTORIAL 1 PARAMETER FOR CUTADAPT (EXTRACTED) Is there any reason to do it in different ways ? I noticed that one of the tutorials using Cutadapt inserts adaptor sequence in "Read 1 Options’’, where as the another tutorial does not, which makes me a bit confused. Hello, I am new to RNA-seq analysis and is following some of the tutorials to analysing my RNA sequences for DGE analysis.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
